In the field of ppb-level NO2 room temperature detection and practical application, significant advancements have been achieved by the research team led by Associate Professor Zhang Xuemin from the College of Sciences, NEU. The scholarly article titled "Sodium titanate: Aproton conduction material for ppb-level NO2 detection with near-zero power consumption" was recently published in the Journal of Hazardous Materials, a reputable publication within the field of environmental engineering. Cai Lubing, a student of class 2002 majoring in applied chemistry in the School of Sciences, NEU, is the first author of the paper. NEU is the only completion unit, with Associate Professor Zhang Xuemin serving as the corresponding author. Cai Lubing has been admitted into NEU's undergraduate to Ph.D. through-class program and has been granted admission as a graduate student in the research group led by Associate Professor Zhang Xuemin in 2024.

The Journal of Hazardous Materials is a scholarly publication affiliated with Elsevier Publishing House. Established in 1975, it holds a prominent position within the realm of environmental research and engineering on a global scale. This journal holds an influential spot in SCI Area 1 of the Chinese Academy of Sciences, securing the 4th rank out of 55 journals under the environmental engineering area.
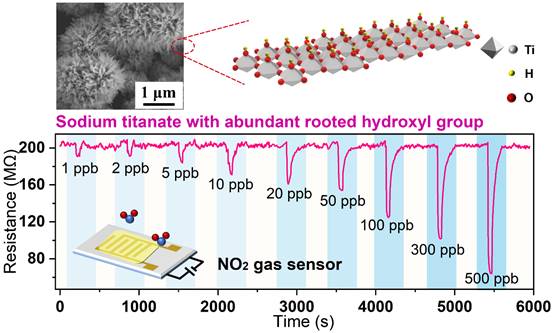
The preparation of NO2 sensors with high sensitivity, fast response/recovery, and low power consumption is a significant challenge due to the limitations imposed by the traditional charge transfer sensing mechanism. In the course of the investigation, sodium titanate (NTO) emerged as a highly promising nitrogen dioxide sensing material. This work aimed to investigate the sensing properties of NTO by examining the influence of operating temperature, applied voltage, and relative humidity. Additionally, a novel sensing mechanism based on NTO surface-bonded hydroxyl groups (OHR) was proposed to explain the observed proton conduction. The primary mechanism of the sensing process primarily entails the preferential adsorption of nitrogen dioxide (NO2) on the oxygen vacancy sites of the oxide heterostructure (OHR). This adsorption event subsequently lowers the activation energy required for the migration of protons along the surface of the nitrogen trioxide (NTO). Simultaneously, the presence of modest intermolecular contacts renders NO2 readily adsorbable and desorbable at ambient conditions. Therefore, NTO has high sensitivity, fast and fully recoverable response (1ppm NO2 response value is 5.7, response time < 3s), wide detection range (1ppb - 20 ppm), good stability (> 2 months) and near-zero power consumption (0.5 nW). Based on the findings, the Nitrogen Trioxide (NO2) sensing capabilities of NTO have been observed to be effective in both indoor and outdoor conditions. The study presents an innovative proton conduction method for the effective detection of NO2, utilizing NTO surface-bonded hydroxyl groups. Additionally, it offers valuable insights for the development and fabrication of sensors targeting various toxic and hazardous gases.
The research conducted in this study received significant financial assistance from the National Natural Science Foundation of China, the Open Project of the State Key Laboratory of Supramolecular Materials and Structures, and the Testing Center of NEU.